How Do You Know if Something Rotates Plane Polarized Light
5.iv Optical Activity
The two enantiomers are mirror images of each other. They are very alike and share many backdrop in common, like same b.p., grand.p., density, color, solubility etc. In fact, the pair of enantiomers have the same physical properties except the way they interact with plane-polarized lite.
In normal low-cal, the electric filed oscillates in all directions. When normal low-cal passed through a polarizing filter, only light oscillating in one unmarried plane tin go through, and the resulting low-cal that oscillates in one single direction is called plane-polarized low-cal.
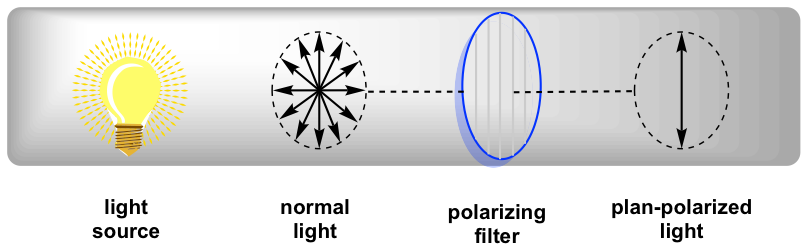
When plan-polarized calorie-free interacts with chiral molecules, the plane of polarization will be rotated by the chiral substances. It was showtime discovered by Jean-Baptiste Biot in 1815 that some naturally occurring organic substances, like camphor, is able to rotate the plane of polarization of plane-polarized light. He likewise noted that some compounds rotated the plane clockwise and others counterclockwise. Farther studies indicate that the rotation is acquired past the chirality of substances.
The property of a chemical compound being able to rotate the plane of polarization of airplane-polarized light is called the optical activity, and the chemical compound with such activity is labelled as optical active. The stereoisomer that is optical agile is also called equally optical isomer.
Chiral compound is optical agile. Achiral chemical compound is optical inactive.
The sample containing a chiral compound rotates the plane of polarization of plane-polarized lite, the management and angles of the rotation depends on the nature and concentration of the chiral substances. The rotation angles can be measured by using polarimeter (later in this section).
For a pair of enantiomers with aforementioned concentration, under the same condition, they rotate the airplane of polarization with the same anglesbut in reverse direction, one is clockwise and the other is counterclockwise.
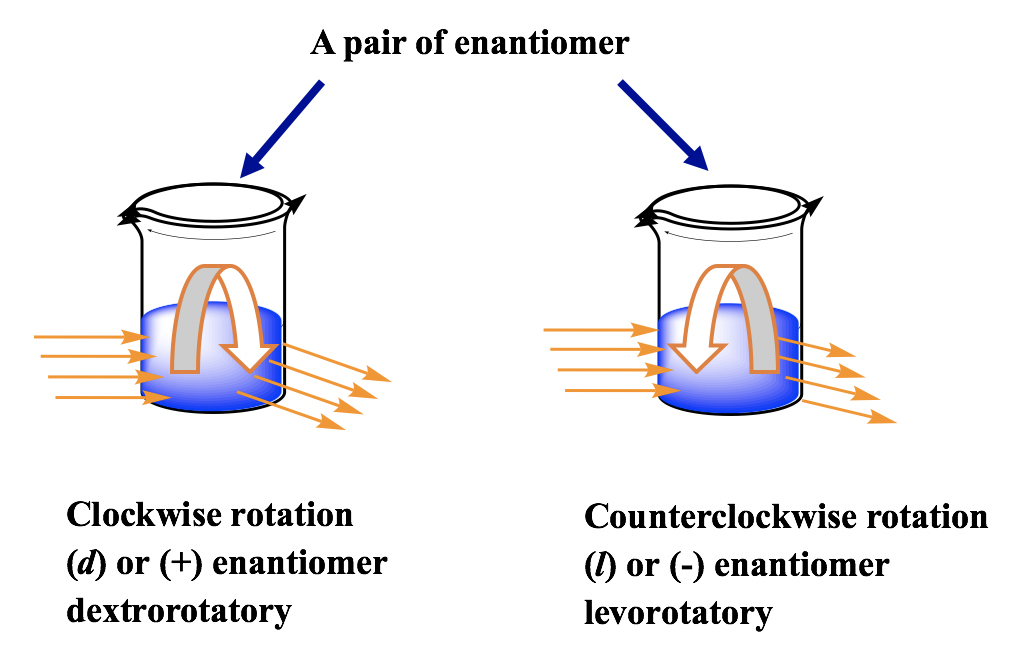
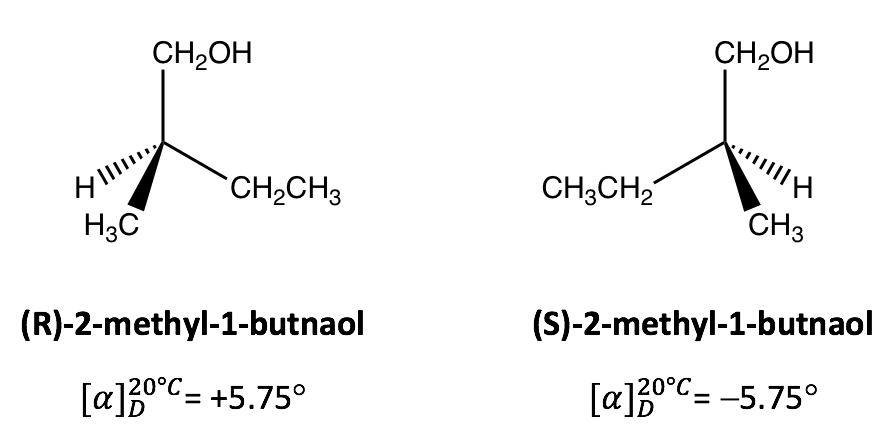
The enantiomer rotates the airplane of polarization clockwise is said to be dextrorotatory(Latin, means to the right), and labelled with the prefix ( d ) or (+). The enantiomer rotates the plane of polarization counterclockwise is said to be levorotatory(Latin, means to the left), and labelled with the prefix ( l ) or (–). The d/l (or +/-) indicate the management in which an optical agile compound rotates the plane of polarization of programme-polarized calorie-free, that has to exist adamant by experiment to measure the optical rotation. d/l (or +/ – ) symbol has nothing to do with R/S. R/S indicates the arrangement of the groups around the chirality center, that tin exist adamant past knowing the exact spatial organization of the groups. That means compound with Rconfiguration can be either dor 50 , and compound with South configuration tin also be either dor l . For the examples beneath, both compounds are South -isomer, just one is d (+)and the other is l (-).

The simply affair we tin be sure is that for a pair of enantiomers, if one enantiomer has been determined as d , and so the other enantiomer must be 50 , and vice versa.
Measurement of Optical Rotation
Polarimeter is the instrument that measures the direction and angles of rotation of plane-polarized light. The plane-polarized lite laissez passer through the sample tube containing the solution of sample, and the angle of rotations will be received and recorded by the analyzer, as summarized in Fig. 5.4c.
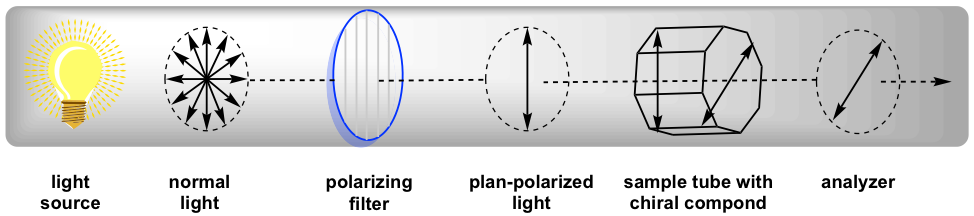
Since the measurement results vary with the wavelength of the light existence used, the specific calorie-free from a sodium atomic spectrum with the wavelength of 589 nm, which is called the sodium D-line, is used for most polarimeter. The rotation degree measured by the polarimeter is called the observed rotation(α), and the observed rotation depends on the length of the sample tube, concentration of the sample and temperature.
To compare the optical rotation betwixt unlike compounds nether consistent conditions, the specific rotation ![]() is used. Specific rotation is the rotation acquired by a solution with concentration of 1.0 g/mL in a sample tube of 1.0 dm length. The temperature is normally at 20°C. Based on this definition, the specific rotation tin exist calculated from the observed rotation past applying the formula:
is used. Specific rotation is the rotation acquired by a solution with concentration of 1.0 g/mL in a sample tube of 1.0 dm length. The temperature is normally at 20°C. Based on this definition, the specific rotation tin exist calculated from the observed rotation past applying the formula:

Please note: In this formula, the unit of concentration (1000/mL) and length of the sample tube (dm) are not the units we are familiar with. Also, the unit of measurement of the specific rotation is in degree (°), don't need to worry about the units cancellation in this formula.
The observed rotation of x.0g of ( R )-ii-methyl-1-butnaol in 50mL of solution in a 20-cm polarimeter tube is +two.three° at twenty °C, what is the specific rotation of the compound?
Solution:
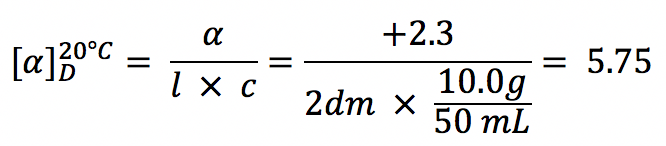
Specific rotation is the feature property of an optical active compound. The literature specific rotation values of the authentic compound can be used to confirm the identity of an unknown compound. For the example here, if it has been measured that the specific rotation of ( R )-2-methyl-1-butnaol is +v.75 ° , then we can tell that the other enantiomer ( S )-2-methyl-1-butnaol must have the specific rotation of -five.75 ° , without further measurement necessary.
Optical Activity of Unlike Samples
When a sample under measurement only contain i enantiomer, this sample is called as enantiomerically pure, means but 1 enantiomer is nowadays in the sample.
The sample may likewise consists of a mixture of a pair of enantiomers. For such mixture sample, the observed rotation value of the mixture, together with the information of the specific rotation of one of the enantiomer permit us to summate the percentage (%) of each enantiomer in the mixture. To do such calculation, the concept of enantiomer backlog (ee) volition be needed. The enantiomeric backlog (ee) tells how much an excess of one enantiomer is in the mixture, and it can be calculated as:
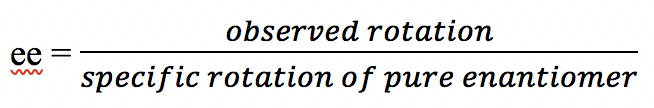
We will use a series of hypothetic examples in adjacent tabular array for detailed explanation.
| If the specific rotation of a (+)-enantiomer is +100 °, so the observed rotation of the post-obit samples are (assume the sample tube has the length of ane dm, and the concentration for each sample is i.0 chiliad/mL): | ||
| Sample Number | Sample | Observed rotation (º) |
| one | pure (+) enantiomer | +100 |
| 2 | Pure (-)-enantiomer | -100 |
| 3 | Racemic mixture of 50% (+)-enantiomer and 50% (-)-enantiomer | 0 |
| iv | Mixture of 75% (+)-enantiomer and 25% (-)-enantiomer | +fifty |
| 4 | Mixture of 20% (+)-enantiomer and 80% (-)-enantiomer | -60 |
Sample #1 and #2 are straightforward.
Sample #3 is for a mixture with equal amount of two enantiomers, and such mixture is called racemic mixtureor racemate. Racemic mixtures do not rotate the plane of polarization of plane-polarized light, that means racemic mixtures are optical inactive and have the observed rotation of zero! This is considering that for every molecule in the mixture that rotate the plane of polarization in one direction, in that location is an enantiomer molecule that rotate the plan of polarization in the opposite direction with the same angle, and the rotation get cancelled out. As a net outcome, no rotation is observed for the overall racemic mixture. The symbol (±) sometimes is used to indicate a mixture is racemic mixture.
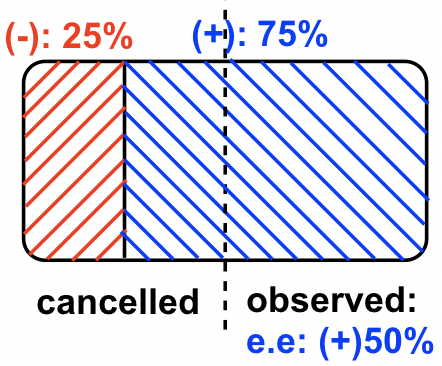
Sample #four, the (+)-enantiomer is in excess. Since there are 75% (+)-enantiomer and 25%(-)-enantiomer, the enantiomeric backlog (ee) value of (+)-enantiomer is 75% – 25% = 50%, this can also exist calculated by the formula: ee = ![]()
In this sample of mixture, the rotation of the (-)-enantiomer is cancelledby the rotation acquired by part of the (+)-enantiomer, so the overall net observed rotation depends on how much "net amount" of (+)-enantiomer present.This can be shown by the diagram below that helps to sympathize.
Sample #5, the (-)-enantiomer is in excess, and because at that place is 80% (-)-enantiomer and xx% (+)-enantiomer, the enantiomeric excess (ee) value of (-)-enantiomer is fourscore% – 20% = 60%, this can also be calculated by the formula: ee = ![]()
Please note:to calculate the e.e value, it is non necessary to include the sign of the rotation bending, every bit long as go on in mind that the sign (+ or –) of the observed rotation indicates that which enantiomer is in excess.
Draw the diagrathousand for Sample #5 by referring to the diagram for Sample #four .
The (+)-enantiomer of a compound has specific rotation ([α]twenty D) of +100°. For a sample (1 one thousand/ml in 1dm prison cell) that is a mixture of (+) and (-) enantiomers, the observed rotation α is -45°, what is the percentage of (+) enantiomer nowadays in this sample?
Solution:
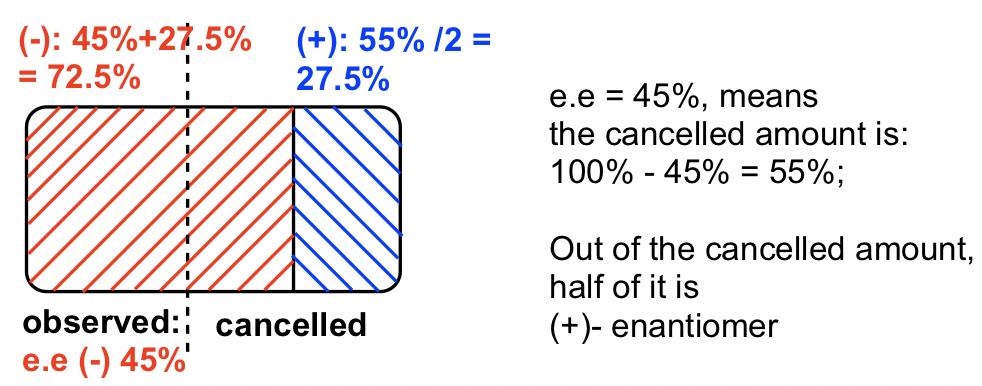
The observed rotation is in "-", so (-)-enantiomer is in excess.
ee of (-)-enantiomer is: ![]()
From here, we will come across two ways of solving such type of question:
Method I : solving algebra
% of (-)-enantiomer is gear up as "ten"; % of (+)-enantiomer is prepare every bit "y"
x + y = 100%
x – y = 45%
Solve x = 72.5%; y = 27.5%;
And then at that place is 72.five% (-)-enantiomer and 27.5% of (+)-enantiomer in the sample.
Method II : using diagram, the answer is in blue color, there is 27.five% of (+)-enantiomer.
Chirality and Biological Properties
Other than optical activity deviation, the dissimilar enantiomers of a chiral molecule usually show different properties when interacting with other chiral substances. This can be understood by using the counterpart example of fitting a hand into the respective glove: right hand only fits into right glove, and information technology feels weird and uncomfortable if you article of clothing left glove on the right hand. This is because both correct hand and correct glove are chiral. A chiral object only fit into a specific chiral environment.
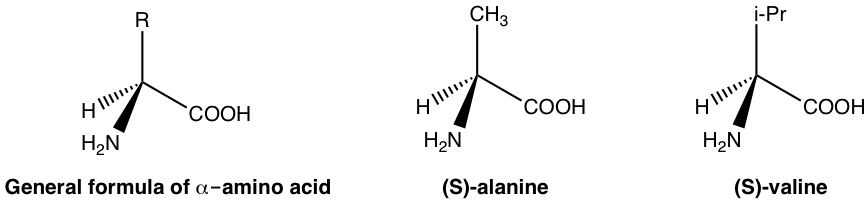
In human torso, the biological functions are modulated by a lot of enzymes and receptors. Enzymes and receptors are essentially proteins, and proteins are made up of amino acids. Amino acids are examples of naturally exist chiral substances. With the full general formula given below, the carbon with amino (NHii) grouping is the chirality (asymmetric) middle for well-nigh amino acids, and only one enantiomer (unremarkably S -enantiomer) exist in nature. A few examples of amino acids are given below with the general formula.
Considering amino acids are chiral, proteins are chiral so enzymes and receptors are chiral as well. The enzyme or receptor therefore form the chiral environs in man body that distinguish between R or Southward enantiomer. Such selectivity can exist illustrated by the simple diagram below.
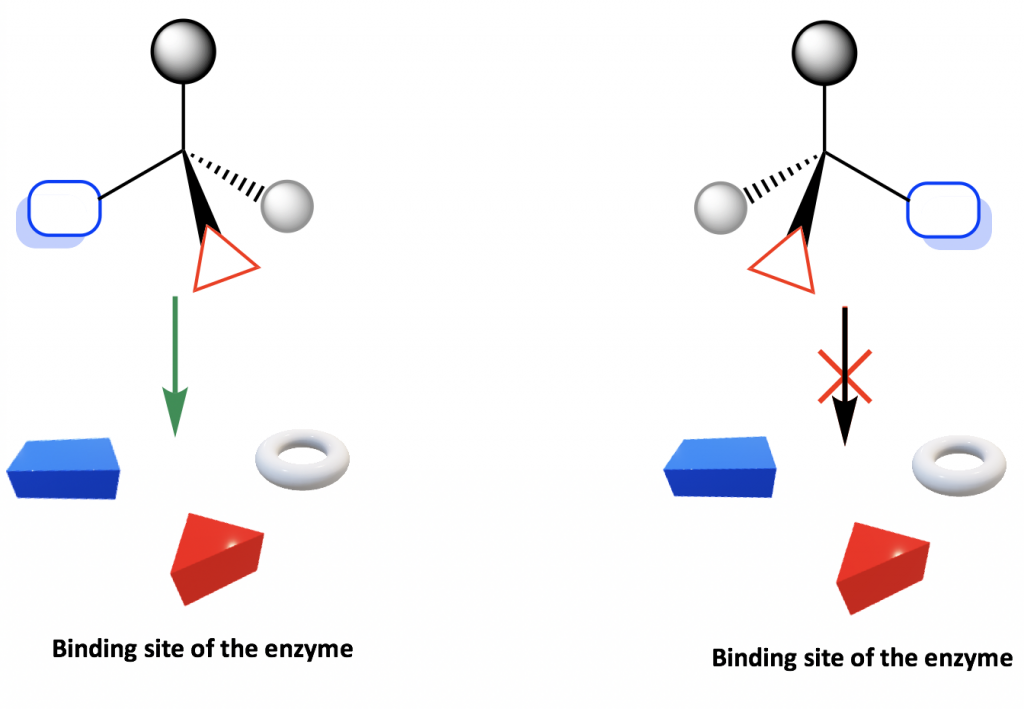
The binding site of enzyme or receptor is chiral, so it only binds with the enantiomer whose groups are in the proper positions to fit into the bounden site. Every bit shown in the diagram, only one enantiomer binds with the site, but non the other enantiomer.
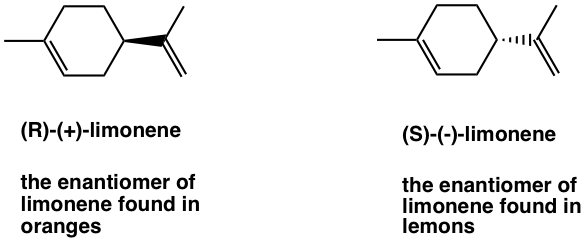
A couple of common examples to showcase such binding selectivity of different enantiomer may include limonene and carvone.
Limonene has two enantiomers, and they smell totally different to human existence because they collaborate with different receptors that located on the nerve cells in nose. The ( R )-(+)-limonene is responsible for the smell of orange, and the ( Southward )-(-)-limonene gives the bit aroma of lemon.
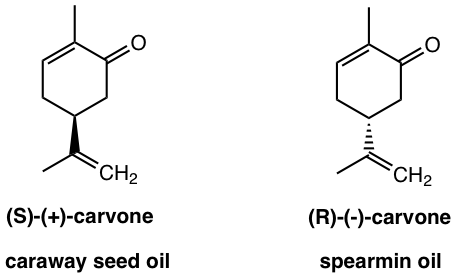
If you like caraway bread, that is due to the ( S )-(+)-carvone; and the ( R )-(-)-carvone that is establish in spearmint oil gives much different odor.
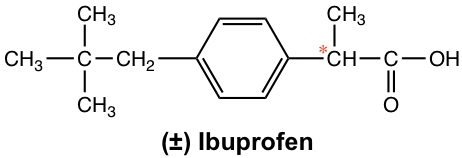
More dramatic examples of how chirality plays important role in biological backdrop are found in many medicines. For the mutual over-counter anti-inflammatory drug ibuprofen (Advil), for instance, merely ( S )-enantiomer is the active agent, while the ( R )-enantiomer has no any anti-inflammatory activeness. Fortunately, the ( R )-enantiomer does not have any harmful side consequence and slowly converts to the ( S )-enantiomer in the trunk. The ibuprofen is marketed usually as a racemate grade.
The issue of chiral drugs (the drug contain a single enantiomer, not as a racemate) was not in the attention of drug discovery industry until 1960. Dorsum then, drugs were approved in racemate grade if a chirality heart involved, and there was no farther written report virtually biological difference on different enantiomers. These were all changed by the tragic incident of thalidomide. Thalidomide was a drug that was sold in more 40 countries, mainly in Europe, in early 1960s equally a sleeping aid and to significant women as antiemetic (drug that preventing vomiting) to combat morning sickness. It was not recognized at that fourth dimension that only the R -enantiomer has the property, while the South -enantiomer was a teratogen that causes congenital deformations. The drug was marketed as a racemic mixture and caused about ten,000 children had been damaged until it was withdrawn from the market in Nov. 1961. This drug was not approved in United states nonetheless, attributed to Dr. Frances O. Kelsey, who was a physician for the FDA (Food and Drug Administration) at that time and had insisted on addition tests on some side effects. Thousands of life were saved by Dr. Kelsey, and she was awarded the President'southward medal in 1962 for preventing the sale of thalidomide.

Source: https://kpu.pressbooks.pub/organicchemistry/chapter/5-3-chirality/#:~:text=The%20d%2Fl%20(or%20%2B%2F%2D,to%20do%20with%20R%2FS.
0 Response to "How Do You Know if Something Rotates Plane Polarized Light"
Post a Comment